
آخر المواضيع المضافة


 الفيزياء الكلاسيكية
الفيزياء الكلاسيكية
 الكهربائية والمغناطيسية
الكهربائية والمغناطيسية
 علم البصريات
علم البصريات
 الفيزياء الحديثة
الفيزياء الحديثة
 النظرية النسبية
النظرية النسبية
 الفيزياء النووية
الفيزياء النووية
 فيزياء الحالة الصلبة
فيزياء الحالة الصلبة
 الليزر
الليزر
 علم الفلك
علم الفلك
 المجموعة الشمسية
المجموعة الشمسية
 الطاقة البديلة
الطاقة البديلة
 الفيزياء والعلوم الأخرى
الفيزياء والعلوم الأخرى
 مواضيع عامة في الفيزياء
مواضيع عامة في الفيزياء|
Read More
Date: 9-12-2020
Date: 30-12-2016
Date: 18-5-2016
|
The First Law of Thermodynamics
We have already studied this! The first law of thermodynamics is nothing more than a re-statement of the work energy theorem, which was
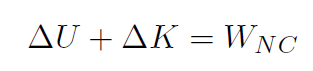
Recall that the total work W was always W = ΔK. Identify heat Q as Q ≡ WNC and internal energy (such as energy stored in a gas, which is just potential energy) is Eint ≡ U and we have
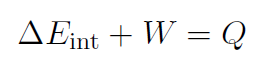
or

which is the first law of thermodynamics. The meaning of this law is that the internal energy of a system can be changed by adding heat or doing work. Often the first law is written for tiny changes as
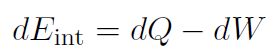



|
|
|
|
4 أسباب تجعلك تضيف الزنجبيل إلى طعامك.. تعرف عليها
|
|
|
|
|
|
|
أكبر محطة للطاقة الكهرومائية في بريطانيا تستعد للانطلاق
|
|
|
|
|
|
|
العتبة العباسية المقدسة تناقش تحديد الرؤية البصرية الخاصة بحفل تخرج طلبة الجامعات العراقية
|
|
|