


 النبات
النبات
 الحيوان
الحيوان
 الأحياء المجهرية
الأحياء المجهرية
 علم الأمراض
علم الأمراض
 التقانة الإحيائية
التقانة الإحيائية
 التقنية الحيوية المكروبية
التقنية الحيوية المكروبية
 التقنية الحياتية النانوية
التقنية الحياتية النانوية
 علم الأجنة
علم الأجنة
 الأحياء الجزيئي
الأحياء الجزيئي
 علم وظائف الأعضاء
علم وظائف الأعضاء
 الغدد
الغدد
 المضادات الحيوية
المضادات الحيوية|
Read More
Date: 7-3-2021
Date: 8-5-2016
Date: 23-12-2015
|
Hydrophilicity
When biological molecules combine or react with each other, or change shape, their interactions with the solvent change. The noncovalent binding of ligands by enzymes, antibodies, and other receptors, eg, involve removal of the interacting partners from the solvent to which they were previously exposed; also, the ease of breaking and making chemical bonds depends on the strength of interaction of the reactants and products with solvent. Most biological molecules contain electronegative atoms such as O, N, or S, which can act as hydrogen bond acceptors or donors and allow them to interact more or less strongly with solvent water; they are said to be hydrophilic . The rates and equilibria of most biological processes, including binding interactions and metabolic transformations, are therefore strongly influenced by changing interactions with the surrounding solvent, which typically is ~80% water in intracellular fluid. Many biological compounds are so polar that their removal from solvent water, an operation essential to measuring the strength of their solvation by water, or hydrophilicity, is difficult. The very difficulty of measuring hydrophilicity suggests the magnitude of the solvation forces and the potential magnitude of their influence on rates and equilibria. Hydrophilicity can be defined as the dimensionless equilibrium constant for the transfer of a molecule between the dilute vapor phase, in which intermolecular contacts are negligible, to dilute aqueous solution, in which each solute molecule is completely surrounded by solvent water. The concentration is measured in the same units, such as moles/liter, in each phase. Hydrophilic character is simply log10 of the equilibrium constant for transfer from vapor to water (1). Hydrophilicity and hydrophilic character values provide the simplest measure of a molecule's affinity for watery surroundings, since they do not depend on any reference phase in which the molecule interacts with other partners. In that sense, they differ from hydrophobicity, which involves a second solvent that is immiscible with water, and with which the solute molecule interacts more or less strongly. Hydrophilic character also differs from solubility, in which the reference phase is the pure solute in liquid or crystalline form: Such an arbitrary reference is of questionable value if different solute molecules are to be compared against each other.
Hydrophilic character can be determined in some cases by measuring the water solubility of a gas under an atmosphere of known partial pressure, or conversely, by measuring the concentration of a solute in the gas space over an aqueous solution of known concentration (Fig. 1). In more typical cases, of the kind often encountered in biological molecules, the concentration of gas in the vapor phase may be too low to detect directly. In such cases, a large volume of carrier gas (usually argon or nitrogen) is bubbled through an aqueous solution of known concentration, then through an efficient trap, which may be water itself. From the amount of solute collecting in the trap, plotted as a function of the measured volume of carrier gas that has passed through the system, it is a simple matter to calculate the concentration of solute in the vapor phase. Using radioisotope labels, UV spectroscopy, or high-field NMR measurements, it is possible in this way to examine molecules as hydrophilic as peptides (2) or guanidino compounds (3), and to determine water-to-vapor distribution coefficients as low as 10–10. A scale of such equilibrium constants for simple organic compounds in uncharged form is shown in Figure 2. More extreme values are beyond direct measurement. However, there is much evidence to suggest that the free energy of solvation by water tends to be an additive function of a molecule's constituent groups (1), allowing values for complex molecules to be estimated by considering their substituents. From the behavior of methylamine and acetic acid, eg, it can be inferred that the concentration of glycine in the vapor phase, over a 1- M aqueous solution, is approximately 10–14M, even though that value is too low to measure directly.
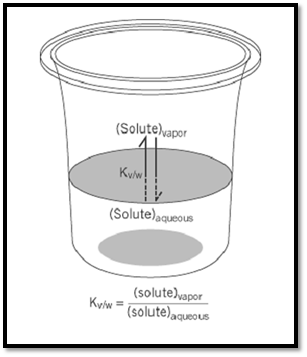
Figure 1. The equilibrium for transfer of a molecule from dilute aqueous solution to the vapor phase. Hydrophilicity is the reciprocal of this equilibrium constant, and it measures the affinity of a molecule for watery surroundings.

Figure 2. Scale of hydrophilicities for simple organic compounds in uncharged form.
These methods have revealed that the hydrophilicities of the protein amino acid side-chains, expressed in terms of their equilibria of transfer from the vapor phase to neutral aqueous solution at 25°C, span a range of approximately 1016-fold(3, 5). These values are as follows:
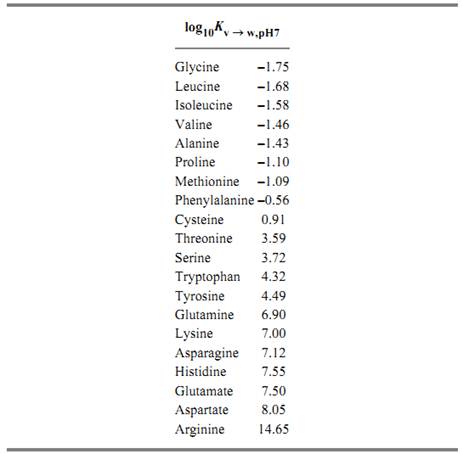
These values were found to be closely related to the tendencies of the corresponding amino acids found at the surfaces of proteins, as revealed by their structures determined by X-ray crystallography. Figure 3 shows the relationship between hydrophilicity and the nominal transfer free energy of each side-chain from the interior to the surface of a protein, as indicated by the relative populations of surface and buried residues for each amino acid. In addition, a remarkable bias in the genetic code was noted, such that purines (usually A) serve as the second code letter for all the more hydrophilic amino acids (3, 4). Figure 4 shows the amino acid side-chains in order of increasing hydrophilicity, alongside the code letters at the second position for each of the amino acids, but the significance of this correlation is not understood.
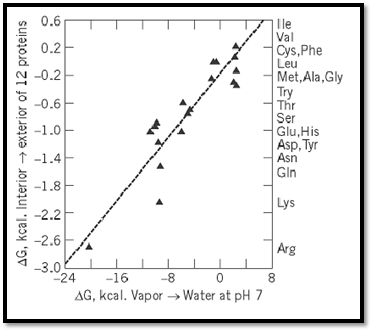
Figure 3. Tendencies of the 20 amino-acid residues to appear at the surfaces of globular proteins, compared with their free energies of transfer from the vapor phase to neutral aqueous solution (3, 4).
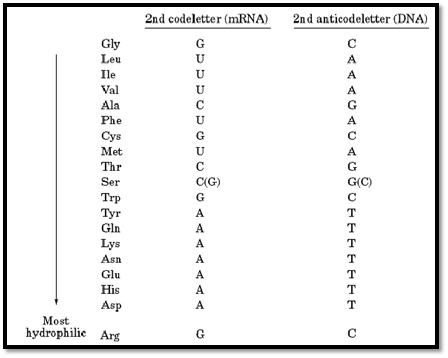
Figure 4. Code letters and anticode letters at the second position for each of the amino acids.
The close relationship observed between hydrophilicity and protein structure (Fig. 3) suggested that this scale might have some value in predicting protein folding. It was also used to propose a
scale of hydropathy values for predicting the relative tendencies of different parts of a protein sequence to be found in membranes (6). More recently, this scale of hydrophilicities has been compared and contrasted with scales of hydrophobicity, obtained by reference to condensed phases including 1- n-octanol and to cyclohexane (7). Completion of a three-sided cycle allows estimation of equilibria of transfer between the vapor phase and cyclohexane, a pure measure of van der Waals forces. As expected, these are found to be roughly proportional to the accessible surface area. Since the interiors of protein molecules represent condensed phases, scales of hydrophobicity can be considered more directly related to folding equilibria than are scales of hydrophilicity using the vapor phase as a reference. Hydrophilicity, on the other hand, is more amenable to theoretical study because it does not entail the involvement of a second condensed phase in addition to water.
References
1. J. Hine and P. K. Mookerjee (1975) J. Org. Chem. 40, 292–298.
2. R. Wolfenden (1978) Biochemistry 17, 201–204.
3. R. Wolfenden, L. Andersson, P. M. Cullis, and C. C. B. Southgate (1981) Biochemistry 20, 849-850.
4. R. Wolfenden, P. M. Cullis, and C. C. B. Southgate (1979) Science 206, 575–577.
5. P. R. Gibbs, A. Radzicka, and R. Wolfenden (1991) J. Amer. Chem. Soc. 113, 4714–4715.
6. J. Kyte and R. F. Doolittle (1982) J. Mol. Biol. 157, 105–132.
7. A. Radzicka and R. Wolfenden (1988) Biochemistry 27, 1664–1670.

|
|
|
|
تفوقت في الاختبار على الجميع.. فاكهة "خارقة" في عالم التغذية
|
|
|
|
|
|
|
أمين عام أوبك: النفط الخام والغاز الطبيعي "هبة من الله"
|
|
|
|
|
|
|
قسم شؤون المعارف ينظم دورة عن آليات عمل الفهارس الفنية للموسوعات والكتب لملاكاته
|
|
|