


 علم الكيمياء
علم الكيمياء 
 الكيمياء التحليلية
الكيمياء التحليلية 
 الكيمياء الحياتية
الكيمياء الحياتية 
 الكيمياء العضوية
الكيمياء العضوية 
 الكيمياء الفيزيائية
الكيمياء الفيزيائية
 الكيمياء اللاعضوية
الكيمياء اللاعضوية 
 مواضيع اخرى في الكيمياء
مواضيع اخرى في الكيمياء
 الكيمياء الصناعية
الكيمياء الصناعية |
Read More
Date: 22-9-2019
Date: 23-10-2019
Date: 21-7-2019
|
Biodegradable Polymers
Plastics derived from natural materials, such as cellulose, starch and hydroxycarboxylic acids are more easily decomposed when exposed to oxygen, water, soil organisms and sunlight than are most petroleum based polymers. The glycoside linkages in polysaccharides and the ester groups in polyesters represent points of attack by the enzymes of microorganisms that facilitate their decomposition. Such biodegradable materials can be composted, broken down and returned to the earth as useful nutrients. However, it is important to recognize that proper composting is necessary. Placing such materials in a landfill results in a slower anaerobic decomposition, which produces methane, a greenhouse gas.
Derivatives of cellulose, such as cellulose acetate, have long served for the manufacture of films and fibers. The most useful acetate material is the diacetate, in which two thirds of the cellulose hydroxyl groups have been esterified. Acetate fibers loose strength when wet, and acetate clothing must be dry cleaned. The other major polysaccharide, starch, is less robust than cellulose, but in pelletized form it is now replacing polystyrene as a packing material.
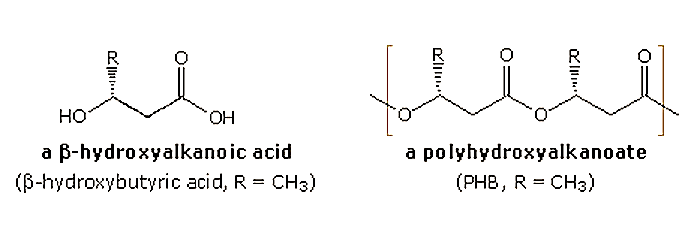
The two natural polyesters that are finding increasing use as replacements for petroleum based plastics are polylactide (PLA) and polyhydroxyalkanoates (PHA), the latter most commonly as copolymers with polyhydroxybutyrate (PHB). Structures for the these polymers and their monomer precursors are shown below.
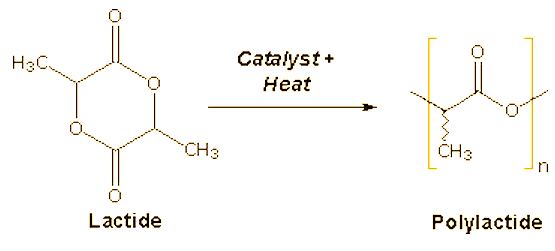
PLA is actually a polymer of lactic acid, but the dimeric lactide is used as the precursor to avoid the water that would be formed in a direct poly-esterification. Bacterial fermentation is used to produce lactic acid from corn starch or cane sugar. After dimerization to the lactide, ring-opening polymerization of the purified lactide is effected using stannous compounds as catalysts. PLA can be processed like most thermoplastics into fibers and films. In situations that require a high level of impact strength, the toughness of PLA in its pristine state is often insufficient. Blends of PLA with polymers such as ABS have good form-stability and visual transparency, making them useful for low-end packaging applications. PLA materials are currently used in a number of biomedical applications, such as sutures, stents, dialysis media and drug delivery devices. However, one of the drawbacks of polylactides for biomedical applications is their brittleness.
Lactic acid has a chiral center, the (S)(+)-enantiomer being the abundant natural form (L-lactic acid). Due to the chiral nature of lactic acid, several distinct forms of polylactide exist. Poly-L-lactide (PLLA) is the product resulting from polymerization of (S,S)-lactide. PLLA has a crystallinity of around 37%, a glass transition temperature between 50-80 ºC and a melting temperature between 173-178 ºC. The melting temperature of PLLA can be increased 40-50 ºC and its heat deformation temperature can be increased from approximately 60 ºC to up to 190 ºC by physically blending the polymer with PDLA (poly-D-lactide). PDLA and PLLA form a highly regular stereocomplex with increased crystallinity.
PHA (polyhydroxyalkanoates) are synthesized by microorganisms such as Alcaligenes eutrophus, grown in a suitable medium and fed appropriate nutrients so that it multiplies rapidly. Once the population has increased, the nutrient composition is changed, forcing the micro-organism to synthesize PHA. Harvested amounts of PHA from the organism can be as high as 80% of the organism's dry weight. The simplest and most commonly occurring form of PHA is poly (R-3-hydroxybutyrate), PHB or P(3HB)). Pure PHB, consisting of 1000 to 30000 hydroxy acid units, is relatively brittle and stiff. Depending upon the microorganism, many of which are genetically engineered for this purpose, and the cultivation conditions, homo- or copolyesters with different hydroxyalkanic acids may be generated. Such copolymers may have improved physical properties compared with homo P(3HB). Presently, these PHAs cost about twice as much as petroleum-based plastics. An engineered switch-grass that grows PHA inside its leaves and stems has also been created, offering the possibility of avoiding some of the costs associated with large scale bacterial fermentation.
In contrast to P(3HB), the polymer of 4-hydroxybutyrate, P(4HB), is elastic and flexible with a higher tensile strength. Copolymers of P(3HB) and P(4HB) are synthesized by Comamonas acidovarans. The molecular weigh remains roughly the same (400,000-700,000 Da), but thermal properties correlate with the ratio of these monomer units. The mp decreases from 179 to 130 (or lower) with an increase in 4HB, and as 4HB increases from 0% to 100% the Tg decreases from 4 to -46. 4-Hydroxybutyrate (4HB) is produced from 1,4-butanediol by microorganisms such asAeromonas hydrophila, Escherichia coli , or Pseudomonas putida. Fermentation broth containing 4HB has then been used for the production of the homopolymer P(4HB), as well as copolymers with P(3HB), [P(3HB-4HB)]. The following table lists some of the properties of these homo-polymers and co-polymers.
|
||||||||||||||||||||||||||||||||||||||||
|
3HV = 3-hydroxyvalerate, 3HD = 3-hydroxydecanoate |
||||||||||||||||||||||||||||||||||||||||



|
|
|
|
تفوقت في الاختبار على الجميع.. فاكهة "خارقة" في عالم التغذية
|
|
|
|
|
|
|
أمين عام أوبك: النفط الخام والغاز الطبيعي "هبة من الله"
|
|
|
|
|
|
|
قسم شؤون المعارف ينظم دورة عن آليات عمل الفهارس الفنية للموسوعات والكتب لملاكاته
|
|
|