


 الفيزياء الكلاسيكية
الفيزياء الكلاسيكية
 الكهربائية والمغناطيسية
الكهربائية والمغناطيسية
 علم البصريات
علم البصريات
 الفيزياء الحديثة
الفيزياء الحديثة
 النظرية النسبية
النظرية النسبية
 الفيزياء النووية
الفيزياء النووية
 فيزياء الحالة الصلبة
فيزياء الحالة الصلبة
 الليزر
الليزر
 علم الفلك
علم الفلك
 المجموعة الشمسية
المجموعة الشمسية
 الطاقة البديلة
الطاقة البديلة
 الفيزياء والعلوم الأخرى
الفيزياء والعلوم الأخرى
 مواضيع عامة في الفيزياء
مواضيع عامة في الفيزياء|
Read More
Date: 8-11-2020
Date: 7-3-2016
Date: 14-7-2020
|
FLUORESCENCE
Fluorescence is the absorption of radiation at one wavelength and emission at a different wavelength. In many cases it is used to convert otherwise useless ultraviolet emissions into useful visible light, as is the case with a fluorescent tube. Fluorescent tubes contain an inert gas with a small amount of mercury in a glass tube. The inert gas (such as argon) at low pressure allows an electrical discharge (started with the help of a glowing filament, which emits electrons) to be sustained through the tube, which in turn excites mercury atoms in the tube by electron collisions. Mercury then emits light; however, most is emitted in the ultraviolet, where it is invisible. A coating of phosphor on the inside wall of the glass tube absorbs UV radiation and reemits this energy in the visible portion of the spectrum as white light. Without this coating of phosphor, the discharge appears to be the characteristic dull blue color characteristic of a mercury discharge, and the UV radiation is simply absorbed by the glass tube and hence wasted. The entire process is depicted in Figure 1.1. By changing the chemical composition of the phosphor,
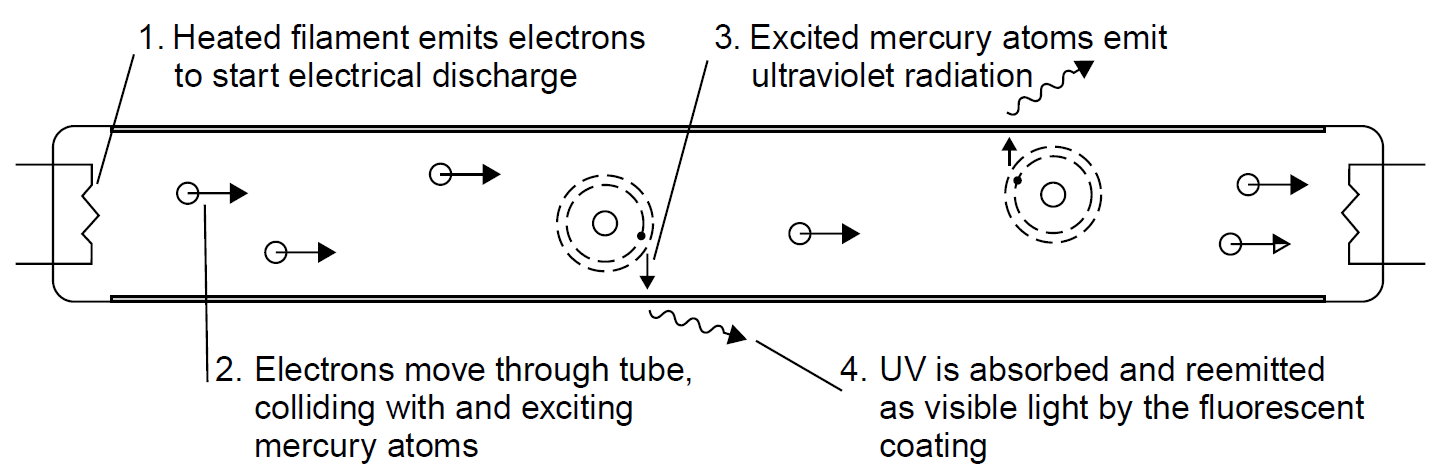
Figure 1.1. Fluorescent tube.
the emission spectrum of the tube can be controlled. Cool white tubes have a phosphor that emits light primarily in the blue-to-yellow region of the spectrum; warm white tubes emit light rich in the orange and red region of the spectrum. The fluorescence process is perhaps most evident in colored neon sign tubes. A pure neon tube containing neon gas glows bright orange, but many modern sign tubes glow blue, pink, green, and many other colors. In most tubes the color is provided by a phosphor coating on the inside of the tube, as evident in Figure 1.2(b). The actual discharge itself is in mercury vapor in a buffer gas of argon. This is the same as a fluorescent tube used for lighting purposes except that neon sign tubes lack a filament to assist starting instead, using high voltages (15 kV is standard for many sign transformers). This discharge in low-pressure mercury vapor produces a large quantity of emission in the ultraviolet. A phosphor coating on the inside of the tube absorbs this UV emission and emits light in the visible spectrum. Phosphors can be formulated to emit light of essentially any color. Fluorescence is also used in vacuum fluorescent displays (VFDs), a popular display technology used on some VCRs and DVD players. In these displays, electrons emitted from a hot filament are accelerated by a relatively low positive voltage of 10 to 15 V through a grid mesh. These electrons then strike anodes which are in the shape of characters or digits. Each anode segment is coated with a phosphor that glows when hit by these electrons. There are numerous anodes, which form segments or dots. When an anode is supplied with a positive voltage relative to the filament (which also serves as a cathode), it will attract the electrons that have been accelerated through the grid. The segment emits light when these electrons strike the phosphor coating. To turn off a segment, anodes are supplied with a negative voltage so that they will repel electrons from their phosphor coating and therefore remain dark. A display of this type is outlined in Figure 1.3. In the figure, both (a) the elements of a VFD and (b) a working display are shown. The filament is visible as well as three lit segments. The filament is overdriven in this case, making it obvious in front of the glowing anodes. In a typical display the filament is heated only to the point where it is barely visible (this provides an adequate source of electrons
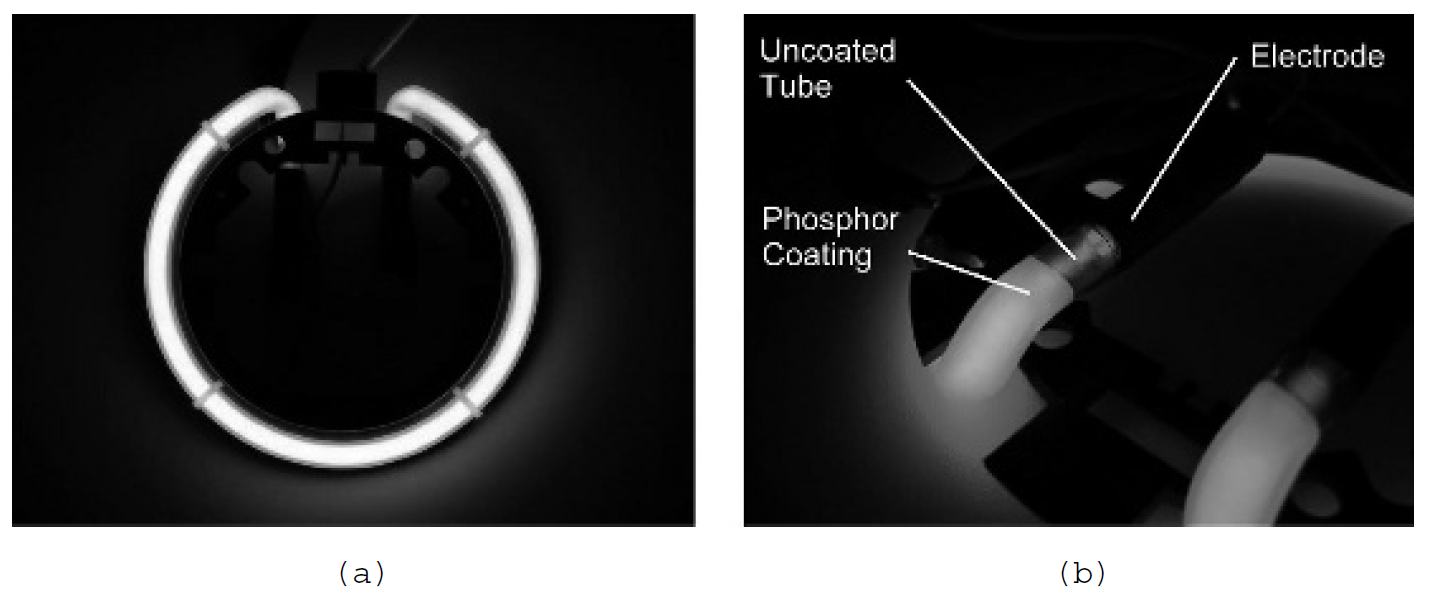
Figure 1.2. Neon sign with phosphor coating.

Figure 1.3. Elements and photo of a vacuum fluorescent display.
to light the anodes) and so is not usually visible, as it is here. Multicolor displays are also possible using this technology; various anodes can be coated with different phosphors to emit light of a desired color. Aside from the obvious applications in fluorescent lamps (including neon tubes) and VFD displays, fluorescence is also used in television picture tubes, where high energy electrons are accelerated from an electron gun in the rear of the tube toward the positively charged front of the tube to strike a phosphor coating, producing light emission. In the case of color television, three phosphors are used, to produce red, green, and blue light, which are mixed to produce any color of light desired, including white.



|
|
|
|
دخلت غرفة فنسيت ماذا تريد من داخلها.. خبير يفسر الحالة
|
|
|
|
|
|
|
ثورة طبية.. ابتكار أصغر جهاز لتنظيم ضربات القلب في العالم
|
|
|
|
|
|
|
العتبة العباسية المقدسة تدعو جامعة ديالى للمشاركة في حفل التخرج المركزي الخامس
|
|
|