


 علم الكيمياء
علم الكيمياء 
 الكيمياء التحليلية
الكيمياء التحليلية 
 الكيمياء الحياتية
الكيمياء الحياتية 
 الكيمياء العضوية
الكيمياء العضوية 
 الكيمياء الفيزيائية
الكيمياء الفيزيائية
 الكيمياء اللاعضوية
الكيمياء اللاعضوية 
 مواضيع اخرى في الكيمياء
مواضيع اخرى في الكيمياء
 الكيمياء الصناعية
الكيمياء الصناعية |
Read More
Date: 1-1-2017
Date: 15-2-2018
Date: 6-2-2018
|
Thermal decompositions of arsine and stibine: the Marsh test
The thermal decomposition of AsH3 and SbH3 is the basis for the Marsh test, which provides a classic example of an analytical technique important in forensic science. The arsenic- or antimony containing material is first converted to AsH3 or SbH3 (e.g. by treatment with Zn and acid, liberating H2 and the trihydride). Subsequent passage of the gaseous mixture through a heated tube causes the hydrides to decompose, forming brown-black deposits of the elements:

If both As and Sb are present, the relative positions of the deposits establishes their identity; the lower thermal stability of SbH3 means that it decomposes before AsH3 in the tube. Treatment of the residues with aqueous NaOCl also distinguishes them since only arsenic dissolves:
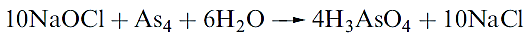



|
|
|
|
التوتر والسرطان.. علماء يحذرون من "صلة خطيرة"
|
|
|
|
|
|
|
مرآة السيارة: مدى دقة عكسها للصورة الصحيحة
|
|
|
|
|
|
|
نحو شراكة وطنية متكاملة.. الأمين العام للعتبة الحسينية يبحث مع وكيل وزارة الخارجية آفاق التعاون المؤسسي
|
|
|