


 علم الكيمياء
علم الكيمياء 
 الكيمياء التحليلية
الكيمياء التحليلية 
 الكيمياء الحياتية
الكيمياء الحياتية 
 الكيمياء العضوية
الكيمياء العضوية 
 الكيمياء الفيزيائية
الكيمياء الفيزيائية
 الكيمياء اللاعضوية
الكيمياء اللاعضوية 
 مواضيع اخرى في الكيمياء
مواضيع اخرى في الكيمياء
 الكيمياء الصناعية
الكيمياء الصناعية |
Read More
Date: 29-6-2020
Date: 23-3-2017
Date: 20-5-2020
|
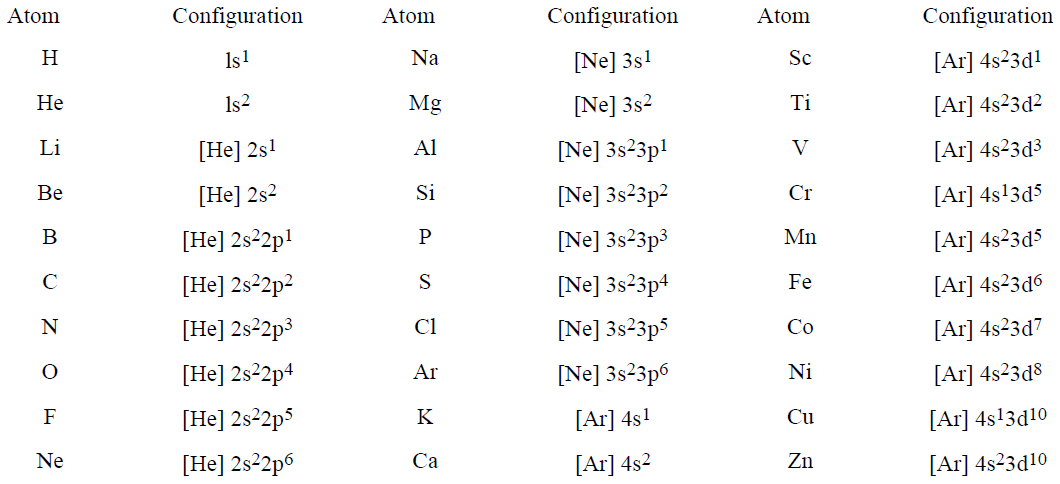
Electronic Configurations
The ground state (the state of lowest energy) of H has a single electron is a 1s orbital, and we use the notation 1s1 to describe the electronic configuration. With 2 electrons, He has the ground state configuration 1s2. When we come to Li with 3 electrons, we must populate the 2s orbital and the ground state configuration is 1s22s1. Table 1.1 shows the ground state configurations for the first 30 elements. In the table, we have used the notation [He], [Ne], and [Ar] to represent the configurations of He, Ne, and Ar; e.g., [Ne] represents the configuration 1s22s22p6. This notation focuses our attention on the outermost electrons, the so-called valence electrons, which are important in chemical bonding.
Table 1.1. Electronic Configurations of Elements 1–30.



|
|
|
|
دراسة: عدم ترتيب الغرفة قد يدل على مشاكل نفسية
|
|
|
|
|
|
|
علماء: تغير المناخ تسبب في ارتفاع الحرارة خلال موسم الحج
|
|
|
|
|
|
|
مركز النبأ العظيم للتربية والتعليم يقيم ورشة عمل حول طرائق التدريس لـ(150) تدريسياً
|
|
|