


 علم الكيمياء
علم الكيمياء 
 الكيمياء التحليلية
الكيمياء التحليلية 
 الكيمياء الحياتية
الكيمياء الحياتية 
 الكيمياء العضوية
الكيمياء العضوية 
 الكيمياء الفيزيائية
الكيمياء الفيزيائية
 الكيمياء اللاعضوية
الكيمياء اللاعضوية 
 مواضيع اخرى في الكيمياء
مواضيع اخرى في الكيمياء
 الكيمياء الصناعية
الكيمياء الصناعية |
Read More
Date: 23-6-2016
Date: 30-10-2019
Date: 9-10-2020
|
Predicting Acid–Base Reactions from pKa Values
Compilations of pKa values like those in Table 1 is useful for predicting whether a given acid–base reaction will take place, because H+ will always go from the stronger acid to the stronger base.
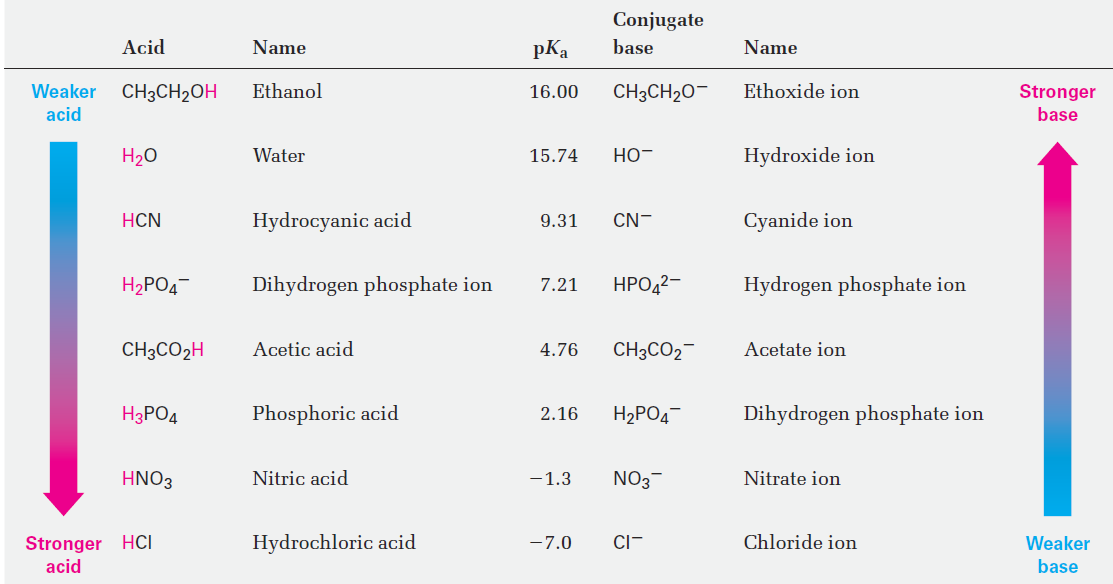
That is, an acid will donate a proton to the conjugate base of a weaker acid, and the conjugate base of a weaker acid will remove a proton from a stronger acid. Since water (pKa = 15.74) is a weaker acid than acetic acid (pKa = 4.76), for example, hydroxide ion holds a proton more tightly than acetate ion does. Hydroxide ion will therefore react to a large extent with acetic acid, CH3CO2H, to yield acetate ion and H2O.

Another way to predict acid–base reactivity is to remember that the product conjugate acid in an acid–base reaction must be weaker and less reactive than the starting acid and the product conjugate base must be weaker and less reactive than the starting base. In the reaction of acetic acid with hydroxide ion, for example, the product conjugate acid (H2O) is weaker than the starting acid (CH3CO2H), and the product conjugate base (CH3CO2-) is weaker than the starting base (OH-).




|
|
|
|
دخلت غرفة فنسيت ماذا تريد من داخلها.. خبير يفسر الحالة
|
|
|
|
|
|
|
ثورة طبية.. ابتكار أصغر جهاز لتنظيم ضربات القلب في العالم
|
|
|
|
|
|
|
العتبة العباسية المقدسة تقدم دعوة إلى كلية مزايا الجامعة للمشاركة في حفل التخرج المركزي الخامس
|
|
|