
آخر المواضيع المضافة


 علم الكيمياء
علم الكيمياء 
 الكيمياء التحليلية
الكيمياء التحليلية 
 الكيمياء الحياتية
الكيمياء الحياتية 
 الكيمياء العضوية
الكيمياء العضوية 
 الكيمياء الفيزيائية
الكيمياء الفيزيائية
 الكيمياء اللاعضوية
الكيمياء اللاعضوية 
 مواضيع اخرى في الكيمياء
مواضيع اخرى في الكيمياء
 الكيمياء الصناعية
الكيمياء الصناعية |
Read More
Date: 27-3-2017
Date: 19-12-2015
Date: 23-8-2016
|
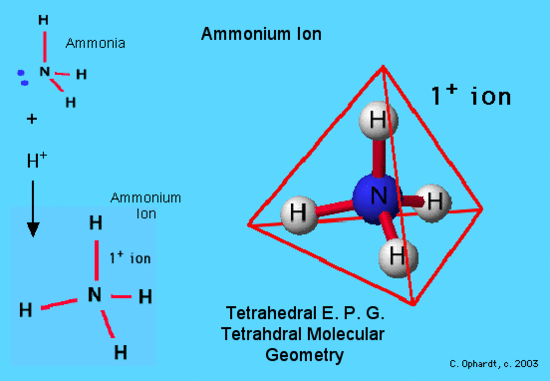
In this example, NH4+, the Lewis diagram shows N at the center with no lone electron pairs. Contrast this with ammonia, NH3, which does have a lone pair. The fourth hydrogen bonds to the ammonia molecule as a hydrogen ion (no electrons) bonding to the lone pair on the nitrogen.
This shows tetrahedral geometry for both the electron pair geometry and the molecular geometry. Ammonium ions are found in solutions of household ammonia or window cleaner. It is also found as part of a solid as ammonium nitrate in fertilizer.



|
|
|
|
"إنقاص الوزن".. مشروب تقليدي قد يتفوق على حقن "أوزيمبيك"
|
|
|
|
|
|
|
الصين تحقق اختراقا بطائرة مسيرة مزودة بالذكاء الاصطناعي
|
|
|
|
|
|
|
قسم شؤون المعارف ووفد من جامعة البصرة يبحثان سبل تعزيز التعاون المشترك
|
|
|