
آخر المواضيع المضافة


 علم الكيمياء
علم الكيمياء 
 الكيمياء التحليلية
الكيمياء التحليلية 
 الكيمياء الحياتية
الكيمياء الحياتية 
 الكيمياء العضوية
الكيمياء العضوية 
 الكيمياء الفيزيائية
الكيمياء الفيزيائية
 الكيمياء اللاعضوية
الكيمياء اللاعضوية 
 مواضيع اخرى في الكيمياء
مواضيع اخرى في الكيمياء
 الكيمياء الصناعية
الكيمياء الصناعية |
Read More
Date: 31-5-2016
Date: 2-11-2019
Date: 27-10-2019
|
Hybrid Orbitals
In order to explain the structure of methane (CH4), the 2s and three 2p orbitals are converted to four equivalent hybrid atomic orbitals, each having 25% s and 75% p character, and designated sp3. These hybrid orbitals have a specific orientation, and the four are naturally oriented in a tetrahedral fashion. Thus, the four covalent bonds of methane consist of shared electron pairs with four hydrogen atoms in a tetrahedral configuration, as predicted by VSEPR theory.
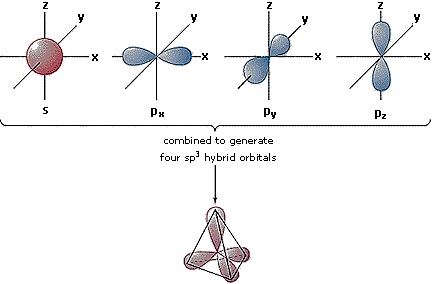



|
|
|
|
أمراض منتصف العمر.. "أعراض خطيرة" سببها نقص المغنيسيوم
|
|
|
|
|
|
|
لماذا تتناقص أعداد النحل عالميا؟
|
|
|
|
|
|
|
ممثل المرجعية العليا: العتبة الحسينية تخطط لإنشاء مراكز متخصصة للكشف المبكر عن (سرطان الثدي) بعموم العراق
|
|
|