


 علم الكيمياء
علم الكيمياء 
 الكيمياء التحليلية
الكيمياء التحليلية 
 الكيمياء الحياتية
الكيمياء الحياتية 
 الكيمياء العضوية
الكيمياء العضوية 
 الكيمياء الفيزيائية
الكيمياء الفيزيائية
 الكيمياء اللاعضوية
الكيمياء اللاعضوية 
 مواضيع اخرى في الكيمياء
مواضيع اخرى في الكيمياء
 الكيمياء الصناعية
الكيمياء الصناعية |
Read More
Date: 25-2-2018
Date: 7-5-2017
Date: 24-4-2017
|
Weight/volume percentage
Weight/volume percentage is very similar to weight/weight percentage, but instead of using grams of solution in the denominator, it uses milliliters of solution:
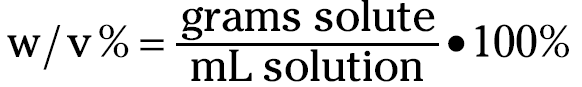
Suppose that you want to make 100 milliliters of a 15 percent (w/v) potassium nitrate solution. Because you’re making 100 milliliters, you already know that you’re going to weigh out 15 grams of potassium nitrate (commonly called saltpeter — KNO3).
Now, here comes something that’s a little different: You’re more concerned with the final volume of the solution than the amount of solvent you use. So you dissolve the 15 grams of KNO3 in a little bit of water and dilute it to exactly 100 milliliters in a volumetric flask. In other words, you dissolve and dilute 15 grams of KNO3 to 100 milliliters. You won’t know exactly how much water you put in, but it’s not important as long as the final volume is 100 milliliters.
You can also use the percentage and volume to calculate the grams of solute present. You may want to know how many grams of sodium hypochlorite are in 500 milliliters of a 5 percent (w/v) solution of household bleach. You can set up the problem like this:

You now know that you have 25 grams of sodium hypochlorite in the 500 milliliters of solution.



|
|
|
|
تفوقت في الاختبار على الجميع.. فاكهة "خارقة" في عالم التغذية
|
|
|
|
|
|
|
أمين عام أوبك: النفط الخام والغاز الطبيعي "هبة من الله"
|
|
|
|
|
|
|
الأمين العام للعتبة العسكرية المقدسة يستقبل قائد الفرقة الرابعة الشرطة الاتحادية
|
|
|