


 علم الكيمياء
علم الكيمياء 
 الكيمياء التحليلية
الكيمياء التحليلية 
 الكيمياء الحياتية
الكيمياء الحياتية 
 الكيمياء العضوية
الكيمياء العضوية 
 الكيمياء الفيزيائية
الكيمياء الفيزيائية
 الكيمياء اللاعضوية
الكيمياء اللاعضوية 
 مواضيع اخرى في الكيمياء
مواضيع اخرى في الكيمياء
 الكيمياء الصناعية
الكيمياء الصناعية |
Read More
Date: 19-9-2018
Date: 12-7-2018
Date: 23-10-2019
|
sp Hybrid Orbitals and the Structure of Acetylene
In addition to forming single and double bonds by sharing two and four electrons, respectively, carbon also can form a triple bond by sharing six electrons. To account for the triple bond in a molecule such as acetylene, H-C≡C-H, we need a third kind of hybrid orbital, an sp hybrid. Imagine that, instead of combining with two or three p orbitals, a carbon 2s orbital hybridizes with only a single p orbital. Two sp hybrid orbitals result, and two p orbitals remain unchanged. The two sp orbitals are oriented 180°apart on the x-axis, while the p orbitals are perpendicular on the y-axis and the z-axis, as shown in Figure 1.

Figure 1 sp Hybridization. The two sp hybrid orbitals are oriented 180° away from each other, perpendicular to the two remaining p orbitals (red/blue).
When two sp-hybridized carbon atoms approach each other, sp hybrid orbitals on each carbon overlap head-on to form a strong sp–sp s bond. At the same time, the pz orbitals from each carbon form a pz–pz π bond by sideways overlap, and the py orbitals overlap similarly to form a py–py π bond. The net effect is the sharing of six electrons and formation of a carbon–carbon triple bond. The two remaining sp hybrid orbitals each form a s bond with hydrogen to complete the acetylene molecule (Figure 2).
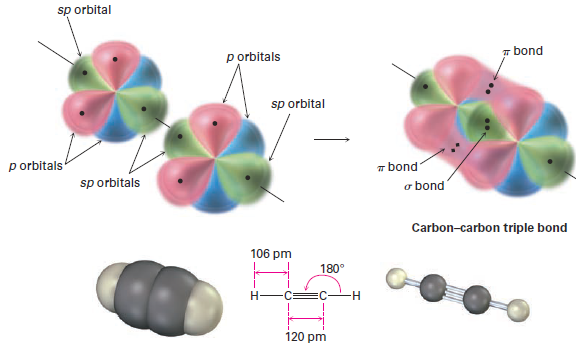
Figure 2 the structure of acetylene. The two carbon atoms are joined by one sp–sp σ bond and two p–p π bonds.
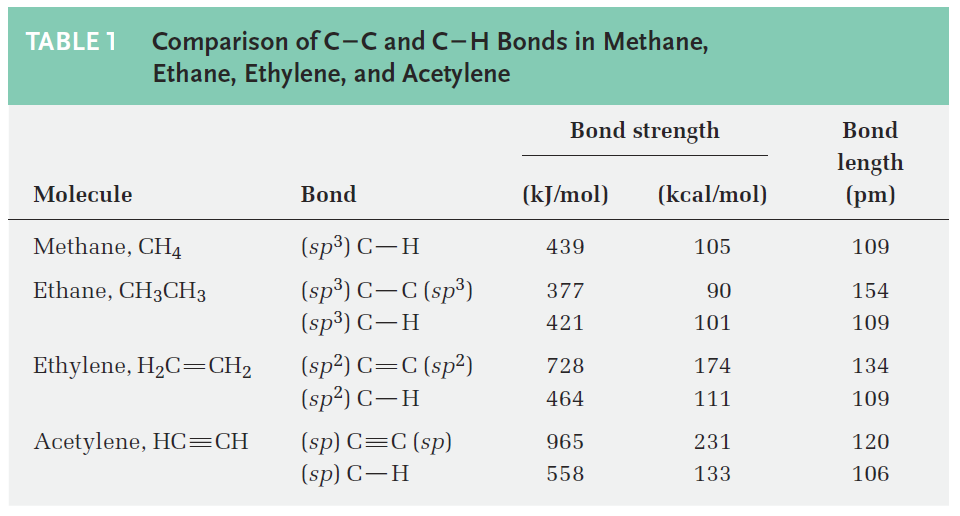
As suggested by sp hybridization, acetylene is a linear molecule with H--C-C bond angles of 180°. The C- H bonds have a length of 106 pm and a strength of 558 kJ/mol (133 kcal/mol). The C-C bond length in acetylene is 120 pm, and its strength is about 965 kJ/mol (231 kcal/mol), making it the shortest and strongest of any carbon–carbon bond. A comparison of sp, sp2, and sp3 hybridization is given in Table 1.



|
|
|
|
دخلت غرفة فنسيت ماذا تريد من داخلها.. خبير يفسر الحالة
|
|
|
|
|
|
|
ثورة طبية.. ابتكار أصغر جهاز لتنظيم ضربات القلب في العالم
|
|
|
|
|
|
|
العتبة العباسية المقدسة تستعد لإطلاق الحفل المركزي لتخرج طلبة الجامعات العراقية
|
|
|