


 النبات
النبات
 الحيوان
الحيوان
 الأحياء المجهرية
الأحياء المجهرية
 علم الأمراض
علم الأمراض
 التقانة الإحيائية
التقانة الإحيائية
 التقنية الحيوية المكروبية
التقنية الحيوية المكروبية
 التقنية الحياتية النانوية
التقنية الحياتية النانوية
 علم الأجنة
علم الأجنة
 الأحياء الجزيئي
الأحياء الجزيئي
 علم وظائف الأعضاء
علم وظائف الأعضاء
 الغدد
الغدد
 المضادات الحيوية
المضادات الحيوية|
Read More
Date: 10-4-2021
Date: 5-5-2016
Date: 3-5-2021
|
Chloramphenicol Acetyltransferase
The enzymic mechanisms by which bacteria modify biologically active molecules are limited by two constraints: (i) the structure–activity correlations for each class of chemical agent and (ii) the metabolic repertoire available to the bacteria. In the case of chloramphenicol (Cml), which has a number of vulnerable functional groups, there are several possibilities. Each functional group of Cml ) Fig. 1) contributes to its effectiveness as an inhibitor of ribosomal peptidyltransferase activity (1), and there are examples (reviewed in Ref. 2) of enzyme-mediated resistance to Cml due to dehalogenation, nitro group reduction, hydrolysis of the amide bond, and modification of the hydroxyl groups by phosphorylation (3) or acetylation (2, 4, 5). Nonetheless, after more than four decades of medical and veterinary use, the preponderant enzymic modification mechanism for Cml resistance in bacteria of clinical importance is that of O-acetylation of the 3-hydroxyl group, catalyzed by chloramphenicol acetyltransferase (CAT) (Fig. 1).

Figure 1. The mechanism of acetylation of the 3-hydroxyl group of chloramphenicol by acetyl-CoA as catalyzed by CAT. The two substrates and the His195 residue of CAT are shown (top). Within the transition state or tetrahedral intermediate (bottom), His195 has abstracted a proton from the 3-hydroxyl group, to generate an “oxyanion” intermediate that has attacked the carbonyl of acetyl-CoA. The intermediate and transition state are stabilized by hydrogen bonding with the side-chain hydroxyl group of Ser148 of CAT.
Genes for CAT are widespread among gram-positive and gram-negative bacteria; the cat gene in each case is either chromosomal or carried by a mobile genetic element, such as a plasmid or transposon. More important biochemically are the properties of representative variants within the CAT “family” and the pattern of conservation of the primary structure, deduced from nucleotide sequences of its genes, which in turn is related to its three-dimensional protein structure, substrate specificity, and catalysis.
All CAT polypeptide chains are in the range 24 to 26 kDa and normally exist in solution as compact and very stable homo-trimers. However, some CAT variants associate in vivo and in vitro to give hybrids, functional a2b and ab2 hetero-trimers with physical and catalytic characteristics reflecting the properties of the parental trimers (6). Because natural isolates of Cml-resistant bacteria may occasionally harbor more than one cat gene, it is possible in such instances that the intracellular CAT pool will include both parental and hybrid trimers.
The extent of structural variation within the CAT family can be appreciated from a comparison of the deduced primary structures for the products of known cat determinants, which yields a lower limit of 28% identity for the most divergent pair of known sequences. Only ~11% of the amino acid residues appear to be identical in all CAT variants, comprising not only residues with side chains that are involved in catalysis and substrate binding, but also those that contribute to critical structural elements necessary for the precise folding and stable packing of the polypeptide chains. A reference point for understanding the variety in primary structures of CAT variants is the type III enzyme
(CATIII), for which a wealth of information is available, including the tertiary structure at high resolution (7, 8) and the structural determinants for the binding of each substrate (2, 4, 5, 7, 8). However, the type I enzyme (CATI) may be the most widely distributed variant, because it is specified by many “F-like” R plasmids of gram-negative bacteria, by transposon Tn9 (9), and by promoter-less cat “cassettes,” constructed in the laboratory for insertion “downstream” of the noncoding sequences of other genes, to study the regulation of their expression. Type II CAT (CATII), notable among “enteric” CATs for its particular sensitivity to inhibition by reagents that react with thiol groups and by its association with Haemophilus influenzae (10), is less commonly encountered. CATI has the remarkable properties of a high affinity for triphenylmethane dyes, such as crystal violet (reviewed in Ref. 2), and the ability to bind a steroidal antibiotic (fusidic acid) both tightly and specifically (11). The latter property is sufficient to confer resistance to fusidate in mutant strains of Escherichia coli that were selected for their sensitivity to the antibiotic prior to the introduction of the gene for CATI. A plausible mechanism is the effective sequestration of fusidate by high levels of CATI, thereby impeding access of the antibiotic to its cellular target, ribosomal elongation factor G. The structural basis for the curious binding of fusidate, which is competitive with respect to Cml, has been deduced by protein engineering and X-ray crystallography (11) . Of the eight residues in the Cml binding pocket of CATIII that differ from those of CATI, only four appear to be responsible for the former's low affinity (Ki = 279 µM) for fusidate. Replacement of each of the four with their counterparts in CATI is sufficient to confer upon CATIII an affinity for fusidate (Ki = 5.4 µM) that approaches that of wild-type CATI (Ki = 1.5 µM).
The catalytic machinery of CATIII comprises a number of amino acid side chains (as well as backbone atoms and ordered water molecules) that make precise contacts with one another or with a substrate molecule. The homo-trimers have rotational symmetry at the molecular threefold axis, and consequently have three identical active sites, so only atoms in a single monomer need be addressed. Nonetheless, each of the structurally and functionally equivalent active sites lies deep in the interfacial clefts between subunits. Central to catalysis (12) is His195, which arises from one face of each cleft to supply the general base (Ne2 in Fig. 1) to deprotonate the C3 hydroxyl of Cml, producing an “oxyanion” intermediate that in turn attacks the carbonyl (C2) carbon of acetyl CoA to yield a tetrahedral intermediate (Fig. 1). Essential for the stabilization of the latter, en route to the transition state for the reaction, and confirmed by site-directed mutagenesis (13), is a negatively charged hydrogen bond (Fig. 1) between the oxyanion and the hydroxyl of Ser148, another residue conserved in all CATs. A neighboring participant in catalysis is Thr174, also conserved, which is hydrogen-bonded to a water molecule that in turn probably makes two hydrogen bonds with the putative tetrahedral intermediate (Fig. 1), one to the 1-hydroxyl of Cml and the other to the 3-oxygen of the intermediate. Two additional conserved residues (Arg18 and Asp199) facilitate catalysis via a network of hydrogen bonds with His195, anchoring the side chain of the latter in a novel conformation that allows it to fulfill its general base role (7, 8, 13-15).
Chloramphenicol becomes acetylated on its 1-hydroxyl group also, albeit at a rate much slower than the 3-acetyl derivative is generated (16). The equations below, all of which are reversible, indicate the transformations involved in the two reactions, indicating that in both cases CAT acetylates only the 3-hydroxyl:

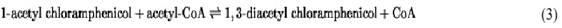
Reaction 2 is a nonenzymic acetyl migration, slow and reversible, which yields at equilibrium a mixture of mono-acetyl products. The 1-acetyl Cml so formed is available for a second round of enzymic acetylation at the C3 position, yielding 1,3-diacetyl Cml. Reaction 3 is ~150-fold less efficient than reaction 1, almost certainly due to an unfavorable “fit” of the substrate at the active site because of the bulky 1-acetyl substituent (17). In any case, the sluggish final step (reaction 3) is of little microbiological significance, because both mono-acetyl derivatives of Cml are already devoid of significant antimicrobial activity, making the rate of reaction 1 the prime determinant of the Cml-resistance phenotype.
In summary, the precise geometry and chemical properties of both substrate binding sites (for Cml and acetyl CoA) and of the catalytic center of CATIII each contribute to its extraordinary efficiency, with a turnover number of 600 s–1 (25°C) with a Km for Cml of 12 µM (~4 µg/mL), reassuringly close to the concentrations at which it inhibits most bacteria of clinical importance. A derived kinetic parameter, the so-called specificity constant (kcat/Km), which combines a measure of substrate affinity with one for catalytic competence, is actually the second-order rate constant for productive collisions of an enzyme with its substrate(s). The value for CATIII and Cml (5 × 107 s–1M–1( approaches that of well-characterized enzyme reactions that are limited by diffusion of the reactants (typically 108 to 109 s–1M–1) and hence at the limit of evolutionary development. By such criteria, CATIII has evolved to a state approaching “perfection” in biological catalysis (18), wherein a fine balance has been struck between rate acceleration (kcat) and specificity (and affinity) for substrate) Km). Although it is not clear how the specificity and catalytic efficiency of CATIII (and related variants) have evolved, the acetyltransferase (E2p) of the pyruvate dehydrogenase complex, which generates acetyl-CoA for central metabolism, has a three-dimensional structure that is virtually identical to that of CAT, as well as the same mechanism of catalysis, but the two proteins have very few identities in primary structure—only those involved with the active site (19).
It is of interest that there is a large family of “xenobiotic” O-acetyltransferases (XATs) with a range of specificities for natural products; several of these enzymes have a low affinity for Cml and hence were first detected as effectors of low-level resistance to the antibiotic (20). All appear to be trimeric but have no sequence homologies with members of the bona fide CAT family described above. One such XAT (with CAT activity) has been studied by X-ray crystallography (21) and shown to have a tertiary structure quite different from that of CAT, but the catalytic mechanism may well involve general base catalysis involving a conserved histidine residue.
References
1. E. F. Gale et al. (1981) The Molecular Basis of Antibiotic Action, 2nd ed., Wiley, London, pp. 462–468.
2. W. V. Shaw (1983) CRC Crit. Rev. Biochem. 14, 1–46.
3. R. H. Mosher et al. (1995) J. Biol. Chem. 27, 27000–27006.
4. W. V. Shaw (1992) Sci. Progress (Oxford) 76, 565–580.
5. W. V. Shaw and A. G. W. Leslie (1991) Annu. Rev. Biophys. Biophys. Chem. 20, 363–386.
6. P. J. Day, I. A. Murray, and W. V. Shaw (1995) Biochemistry 34, 6416–6422.
7. A. G. W. Leslie (1990) J. Mol. Biol. 213, 167–186.
8. A. G. W. Leslie, P. C. E. Moody, and W. V. Shaw (1988) Proc. Natl. Acad. Sci. USA 85, 4133–4137 .
9. N. E. Alton and D. Vapnek (1979) Nature 282, 864–869.
10. I. A. Murray, J. V. Martinez-Suarez, T. J. Close, and W. V. Shaw (1990) Biochem. J. 272, 505–510 .
11. I. A. Murray et al. (1995) J. Mol. Biol. 254, 993–1005.
12. A. Lewendon et al. (1994) Biochemistry 33, 1944–1950.
13. A. Lewendon, I. A. Murray, W. V. Shaw, M. R. Gibbs, and A. G. W. Leslie (1990( Biochemistry 9, 2075–2080.
14. A. Lewendon et al. (1988) Biochemistry 27, 7385–7390.
15. A. Lewendon and W. V. Shaw (1993) J. Biol. Chem. 268, 20997–21001.
16. J. Ellis, C. R. Bagshaw, and W. V. Shaw (1995) Biochemistry 34, 16852–16859.
17. I. A. Murray et al. (1991) Biochemistry 30, 3763–3770.
18. W. J. Albery and J. R. Knowles (1976) Biochemistry 15, 5631–5640.
19. A. Mattevi et al. (1993) Biochemistry 32, 3887–3901.
20. I. A. Murray and W. V. Shaw (1997) Antimicrob. Agents Chemother. 41, 1–6.
21. T. W. Beaman, M. Sugantino, and S. L. Roderick (1998) Biochemistry 37, 6689–6696.

|
|
|
|
4 أسباب تجعلك تضيف الزنجبيل إلى طعامك.. تعرف عليها
|
|
|
|
|
|
|
أكبر محطة للطاقة الكهرومائية في بريطانيا تستعد للانطلاق
|
|
|
|
|
|
|
العتبة العباسية المقدسة تبحث مع العتبة الحسينية المقدسة التنسيق المشترك لإقامة حفل تخرج طلبة الجامعات
|
|
|