


 علم الكيمياء
علم الكيمياء 
 الكيمياء التحليلية
الكيمياء التحليلية 
 الكيمياء الحياتية
الكيمياء الحياتية 
 الكيمياء العضوية
الكيمياء العضوية 
 الكيمياء الفيزيائية
الكيمياء الفيزيائية
 الكيمياء اللاعضوية
الكيمياء اللاعضوية 
 مواضيع اخرى في الكيمياء
مواضيع اخرى في الكيمياء
 الكيمياء الصناعية
الكيمياء الصناعية |
Read More
Date: 5-7-2019
Date: 18-1-2020
Date: 28-8-2018
|
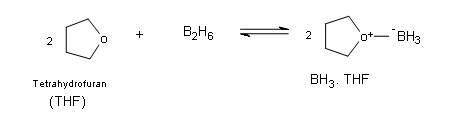
First off it is very imporatnt to understand little bit about the structure and the properties of the borane molecule. Borane exists naturally as a very toxic gas and it exists as dimer of the general formula B2H6 (diborane). Additionally, the dimer B2H6ignites spontaneously in air. Borane is commercially available in ether and tetrahydrofuran (THF), in these solutions the borane can exist as a lewis acid-base complex, which allows boron to have an electron octet.BH3→B2H6
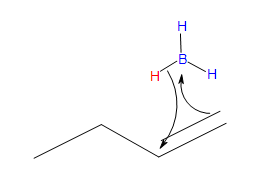
Transition state
.bmp?revision=1&size=bestfit&width=441&height=242)
* Note that a carbocation is not formed. Therefore, no rearrangement takes place.
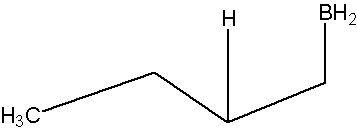

.bmp?revision=1&size=bestfit&width=696&height=239)
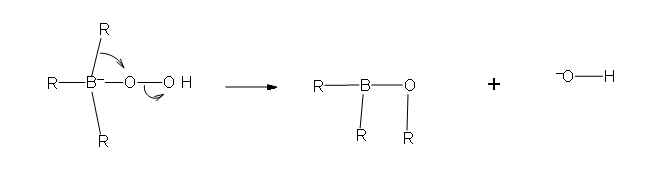
Two more of these reactions with hydroperoxide will occur in order give a trialkylborate
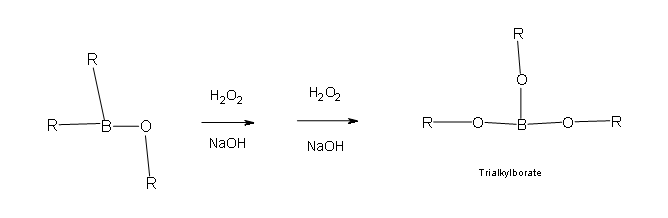

If you need additional visuals to aid you in understanding the mechanism, click on the outside links provided here that will take you to other pages and media that are very helpful as well.



|
|
|
|
دخلت غرفة فنسيت ماذا تريد من داخلها.. خبير يفسر الحالة
|
|
|
|
|
|
|
ثورة طبية.. ابتكار أصغر جهاز لتنظيم ضربات القلب في العالم
|
|
|
|
|
|
|
العتبة العباسية المقدسة تستعد لإطلاق الحفل المركزي لتخرج طلبة الجامعات العراقية
|
|
|