
آخر المواضيع المضافة


 علم الكيمياء
علم الكيمياء 
 الكيمياء التحليلية
الكيمياء التحليلية 
 الكيمياء الحياتية
الكيمياء الحياتية 
 الكيمياء العضوية
الكيمياء العضوية 
 الكيمياء الفيزيائية
الكيمياء الفيزيائية
 الكيمياء اللاعضوية
الكيمياء اللاعضوية 
 مواضيع اخرى في الكيمياء
مواضيع اخرى في الكيمياء
 الكيمياء الصناعية
الكيمياء الصناعية |
Read More
Date: 22-3-2017
Date: 10-5-2020
Date: 25-6-2019
|
Orbital energies in a hydrogen-like species
Besides providing information about the wave functions, solutions of the Schrodinger equation give orbital energies, E (energy levels), and equation 1.16 shows the dependence of E on the principal quantum number for hydrogen-like species.

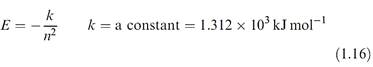
For each value of n there is only one energy solution and for hydrogen-like species, all atomic orbitals with the same principal quantum number (e.g. 3s, 3p and 3d ) are degenerate.



|
|
|
|
التوتر والسرطان.. علماء يحذرون من "صلة خطيرة"
|
|
|
|
|
|
|
مرآة السيارة: مدى دقة عكسها للصورة الصحيحة
|
|
|
|
|
|
|
نحو شراكة وطنية متكاملة.. الأمين العام للعتبة الحسينية يبحث مع وكيل وزارة الخارجية آفاق التعاون المؤسسي
|
|
|