


 علم الكيمياء
علم الكيمياء 
 الكيمياء التحليلية
الكيمياء التحليلية 
 الكيمياء الحياتية
الكيمياء الحياتية 
 الكيمياء العضوية
الكيمياء العضوية 
 الكيمياء الفيزيائية
الكيمياء الفيزيائية
 الكيمياء اللاعضوية
الكيمياء اللاعضوية 
 مواضيع اخرى في الكيمياء
مواضيع اخرى في الكيمياء
 الكيمياء الصناعية
الكيمياء الصناعية |
Read More
Date: 8-1-2020
Date: 12-5-2016
Date: 7-10-2020
|
To resolve a racemic alcohol, a chiral acid can be used to convert the alcohol to a mixture of diastereomeric esters. This is not as generally useful as might be thought because esters tend to be liquids unless they are very high-molecularweight compounds. If the diastereomeric esters are not crystalline, they must be separated by some other method than fractional crystallization (for instance, by chromatography methods, Section 9-2). Two chiral acids that are useful resolving agents for alcohols are:
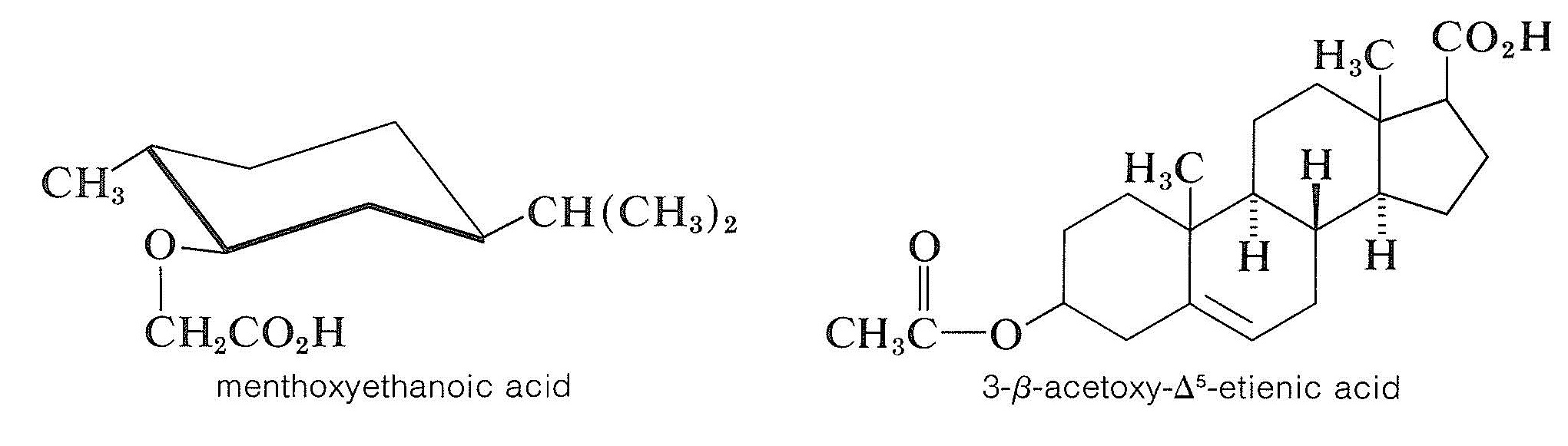
The most common method of resolving an alcohol is to convert it to a half-ester of a dicarboxylic acid, such as butanedioic (succinic) or 1,2-benzenedicarboxylic (phthalic) acid, with the corresponding anhydride. The resulting half-ester has a free carboxyl function and may then be resolvable with a chiral base, usually brucine:
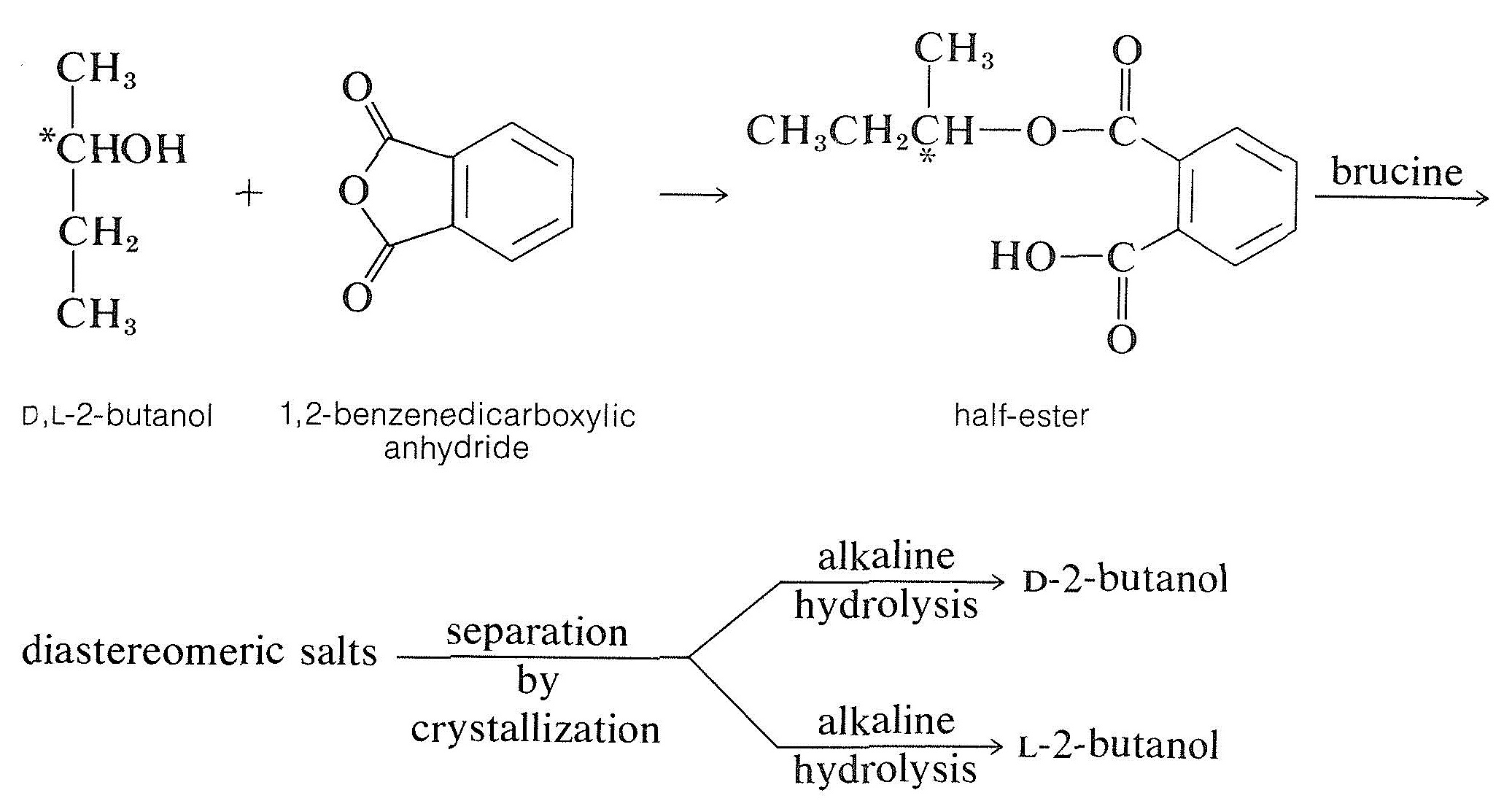



|
|
|
|
4 أسباب تجعلك تضيف الزنجبيل إلى طعامك.. تعرف عليها
|
|
|
|
|
|
|
أكبر محطة للطاقة الكهرومائية في بريطانيا تستعد للانطلاق
|
|
|
|
|
|
|
العتبة العباسية المقدسة تبحث مع العتبة الحسينية المقدسة التنسيق المشترك لإقامة حفل تخرج طلبة الجامعات
|
|
|