


 علم الكيمياء
علم الكيمياء 
 الكيمياء التحليلية
الكيمياء التحليلية 
 الكيمياء الحياتية
الكيمياء الحياتية 
 الكيمياء العضوية
الكيمياء العضوية 
 الكيمياء الفيزيائية
الكيمياء الفيزيائية
 الكيمياء اللاعضوية
الكيمياء اللاعضوية 
 مواضيع اخرى في الكيمياء
مواضيع اخرى في الكيمياء
 الكيمياء الصناعية
الكيمياء الصناعية |
Read More
Date: 21-5-2019
Date: 19-5-2019
Date: 21-5-2019
|
Solutions and Their Concentrations
Over the course of history, measurements and their corresponding units were invented at the local level. By necessity of primitive communication and local technology, standards were nearly nonexistent, and conversions among the many systems were difficult. The result was many hundreds of distinct ways of expressing concentrations of solutions. Fortunately for us, the advent of rapid communications technology and the development of efficient travel have forced globalization of measurement science and, along with it, the definition of global measurement standards. No field has enjoyed more benefit in this regard than chemistry in general and analytical chemistry in particular. Even so, we use a number of methods for expressing concentration.
Concentration of Solutions
In the pages that follow, we describe the four fundamental ways of expressing solution concentration: molar concentration, percent concentration, solution-diluent volume ratio, and p-functions.
Molar Concentration
The molar concentration cx of a solution of a solute species X is the number of moles of that species that is contained in 1 liter of the solution (not 1 L of the solvent).
In terms of the number of moles of solute, n, and the volume, V, of solution, we write
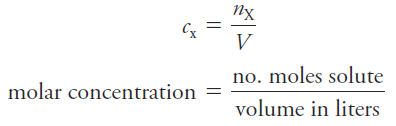
The unit of molar concentration is molar, symbolized by M, which has the dimensions of mol/L, or mol.L-1. Molar concentration is also the number of millimoles of solute per milliliter of solution.
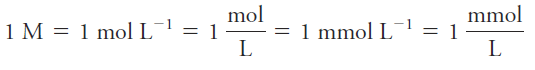




|
|
|
|
لخفض ضغط الدم.. دراسة تحدد "تمارين مهمة"
|
|
|
|
|
|
|
طال انتظارها.. ميزة جديدة من "واتساب" تعزز الخصوصية
|
|
|
|
|
|
|
في يومه الثاني.. حفل التخرج المركزي يشهد إقامة ورشة عن صناعة الطموح
|
|
|