

النبات

مواضيع عامة في علم النبات

الجذور - السيقان - الأوراق

النباتات الوعائية واللاوعائية

البذور (مغطاة البذور - عاريات البذور)

الطحالب

النباتات الطبية


الحيوان

مواضيع عامة في علم الحيوان

علم التشريح

التنوع الإحيائي

البايلوجيا الخلوية


الأحياء المجهرية

البكتيريا

الفطريات

الطفيليات

الفايروسات


علم الأمراض

الاورام

الامراض الوراثية

الامراض المناعية

الامراض المدارية

اضطرابات الدورة الدموية

مواضيع عامة في علم الامراض

الحشرات


التقانة الإحيائية

مواضيع عامة في التقانة الإحيائية


التقنية الحيوية المكروبية

التقنية الحيوية والميكروبات

الفعاليات الحيوية

وراثة الاحياء المجهرية

تصنيف الاحياء المجهرية

الاحياء المجهرية في الطبيعة

أيض الاجهاد

التقنية الحيوية والبيئة

التقنية الحيوية والطب

التقنية الحيوية والزراعة

التقنية الحيوية والصناعة

التقنية الحيوية والطاقة

البحار والطحالب الصغيرة

عزل البروتين

هندسة الجينات


التقنية الحياتية النانوية

مفاهيم التقنية الحيوية النانوية

التراكيب النانوية والمجاهر المستخدمة في رؤيتها

تصنيع وتخليق المواد النانوية

تطبيقات التقنية النانوية والحيوية النانوية

الرقائق والمتحسسات الحيوية

المصفوفات المجهرية وحاسوب الدنا

اللقاحات

البيئة والتلوث


علم الأجنة

اعضاء التكاثر وتشكل الاعراس

الاخصاب

التشطر

العصيبة وتشكل الجسيدات

تشكل اللواحق الجنينية

تكون المعيدة وظهور الطبقات الجنينية

مقدمة لعلم الاجنة


الأحياء الجزيئي

مواضيع عامة في الاحياء الجزيئي


علم وظائف الأعضاء


الغدد

مواضيع عامة في الغدد

الغدد الصم و هرموناتها

الجسم تحت السريري

الغدة النخامية

الغدة الكظرية

الغدة التناسلية

الغدة الدرقية والجار الدرقية

الغدة البنكرياسية

الغدة الصنوبرية

مواضيع عامة في علم وظائف الاعضاء

الخلية الحيوانية

الجهاز العصبي

أعضاء الحس

الجهاز العضلي

السوائل الجسمية

الجهاز الدوري والليمف

الجهاز التنفسي

الجهاز الهضمي

الجهاز البولي


المضادات الميكروبية

مواضيع عامة في المضادات الميكروبية

مضادات البكتيريا

مضادات الفطريات

مضادات الطفيليات

مضادات الفايروسات

علم الخلية

الوراثة

الأحياء العامة

المناعة

التحليلات المرضية

الكيمياء الحيوية

مواضيع متنوعة أخرى

الانزيمات
Chimeric Antibodies
المؤلف:
John M Walker and Ralph Rapley
المصدر:
Molecular Biology and Biotechnology 5th Edition
الجزء والصفحة:
8-12-2020
2456
Chimeric Antibodies
the development of chimeric antibodies was a significant leap forward for the therapeutic antibody field. To reduce the immunogenicity of murine antibodies, the murine variable regions were grafted on to human kappa light chain and gamma heavy chain constant regions . This is relatively straightforward as the V regions and C regions are contiguous pieces of DNA and each domain folds independently.
For chimerization, the DNA encoding the antibody heavy (VH) and light (VL) chain variable region genes must be cloned from the hybridoma cell. Prior to the development of the polymerase chain reaction (PCR), this required cDNA cloning of the variable region genes. With the advent of PCR, 5´ PCR primers were initially designed based on N-terminal protein sequencing of the variable regions from the heavy and light chain. Design of the primer was more straightforward, as it could be based in the constant regions. A major breakthrough was the development of partially degenerate universal V region primers based on sequence databases that could be used to amplify and directionally clone most murine variable regions without the need for protein sequencing (Figure1). Additional groups have designed sets of universal V gene primers containing internal or appended restriction sites suitable for amplification of murine, human,chicken and rabbit V genes. Once the VH and VL genes have been cloned, they can be directly forced into cloning vectors by incorporating restriction sites into the primers. Alternatively, PCR can be used to combine VH and VL genes directly with linker DNA using splicing by overlap extension to generate antigen binding antibody fragment constructs such as those encoding

Figure 1: PCR cloning of antibody V genes. (1) mRNA is isolated from hybridoma cells, peripheral blood lymphocytes, spleen or bone marrow and antibody genes are reverse transcribed (by using reverse transcriptase) using IgG, IgM, k or l constant-region specific primers, creating first strand cDNA. (2) The VH and VL variable region genes are amplified using PCR and universal primer mixtures specific for the 50 (back primers) and 3´ (forward primers) ends of the heavy and light chain V genes. (3a) Amplified VH and VL gene DNA is purified and cloned into cloning vectors for DNA sequencing. By incorporating restriction sites into the primers, it is possible to directionally clone the V genes. (3b) Alternatively, the amplified VH and VL genes can be combined with a short ‘linker DNA’ which overlaps the 3´and 5´ ends of the VH and VL genes, respectively, and PCR amplified to yield one continuous DNA fragment. The linker DNA encodes a flexible peptide that links the VH and VL gene to generate s single-chain Fv (scFv) gene. A final PCR reaction (not shown) adds flanking restriction sites to the assembled scFv gene for cloning into bacterial secretion vectors or display vectors.
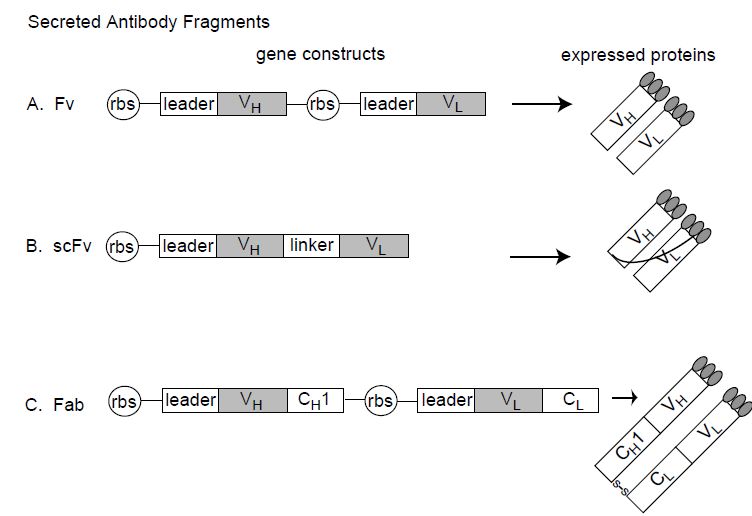
Figure 2: Antibody fragments that can be expressed in E. coli. Fv, single-chain Fv (scFv) and Fab antibody fragments can be expressed in E. coli by directing the V domains into the bacterial periplasm using a leader sequence. rbs=ribosome binding site; leader¼bacterial secretion signal directing the expressed protein to the bacterial periplasm; VH=heavy chain variable domain; VL=ight chain variable domain; linker=flexible scFv linker between VH and VL domains; CH1=heavy chain constant domain 1; CL=light chain constant domain; S–S indicates the disulfide bond between the CH1 and the CL domains.
single chain Fv antibodies (scFv, Figure 1 3b). By incorporating restriction sites into the primers, the scFv genes can be directionally cloned into bacterial secretion vectors (see below and Figure 2), which allow antibody fragment expression and screening for antigen binding. Although PCR greatly simplified the cloning of V genes, mutations introduced by the somatic hypermutation machinery into the regions where the primers anneal may make PCR amplification difficult or impossible, necessitating another amplification approach such as RACE or oligoligation PCR. Cloning the correct VH and VL can also be complicated by the presence of several immunoglobulin transcripts, some of them arising from the fusion partner. In the original examples of chimerization, vectors containing murine V regions fused to human constant regions were constructed and used to transfect mouse myeloma cell lines to produce antibody to confirm antigen binding, affinity and specificity. This process, which ensures that the correct VH and VL genes have been cloned from the hybridoma, takes several months if one constructs stable mammalian cell lines, for example in myeloma cells. An alternative would be to express the chimeric antibody transiently in mammalian cells such as COS-7 cells, cutting the time from cloning to screening for antigen binding to several weeks. Commercially, full-length IgG (chimeric, humanized or human) are typically expressed in Chinese hamster ovary (CHO) cells at yields greater than 1 g l-1.
A major step forward in antibody engineering occurred with the discovery that antibody fragments could be expressed in bacteria, such as E. coli (Figure 2). Due to the rapid growth of bacteria, time from V gene cloning to screening of antibody fragments for binding can as short as several days. It is now common practice after cloning V genes from hybridomas to express them as antibody fragments in bacteria to verify antigen binding prior to construction of chimeric IgG or prior to humanization (see below). Initial attempts to express full-length antibody in the cytoplasm of E. coli resulted in very low yields of insoluble protein that required solubilization and refolding. A significant breakthrough occurred when it was discovered that antigen binding fragments of antibodies could be produced in properly folded soluble form if expression of the antibody fragment was directed into thepe riplasmic space. Secretion into the periplasm was directed by attaching signal sequences such as pelB to the N-terminus of the antibody fragment genes (Figure 2). The oxidizing environment of the periplasm results in proper folding and intramolecular disulfide bond formation.
Both the Fab and the Fv antigen binding fragments can be expressed in E. coli where they can be harvested from the bacterial periplasm. Fvs, however, are not particularly stable; the VH and VL domains are not covalently linked and they tend to dissociate at typically expressed concentrations. Fvs can be stabilized by physically linking the VH and VL domains together with a flexible peptide linker to create the single chain Fv antibody fragments (scFv).Alternatively, cysteines can be engineered into both the VH and VL domains, resulting in disulfide bond formation when the V domains pair, a so-called disulfide linked Fv.
A number of chimeric antibodies have been approved by the FDA and entered clinical practice, including rituximab and infliximab. Chimerization reduces the immunogenicity of murine mAbs and allows multiple and repeated dosing.Replacing the murine Fc with a human Fc also results in more efficient effector functions such as ADCC and CDC as the human Fc interacts with human Fc receptors with higher affinity than the murine Fc. Similarly, chimeric antibodies have a longer half-life in humans than murine antibodies, due to more efficient interaction of the human Fc with FcRn. Despite their successes, however, chimeric antibodies can still be immunogenic. A human anti-chimeric antibody (HACA) response is frequently observed and in some cases can be severe and require discontinuation of the antibody or result in its ineffectiveness. As a result, techniques to generate more fully human antibodies were developed.
 الاكثر قراءة في المناعة
الاكثر قراءة في المناعة
 اخر الاخبار
اخر الاخبار
اخبار العتبة العباسية المقدسة

الآخبار الصحية















 قسم الشؤون الفكرية يصدر كتاباً يوثق تاريخ السدانة في العتبة العباسية المقدسة
قسم الشؤون الفكرية يصدر كتاباً يوثق تاريخ السدانة في العتبة العباسية المقدسة "المهمة".. إصدار قصصي يوثّق القصص الفائزة في مسابقة فتوى الدفاع المقدسة للقصة القصيرة
"المهمة".. إصدار قصصي يوثّق القصص الفائزة في مسابقة فتوى الدفاع المقدسة للقصة القصيرة (نوافذ).. إصدار أدبي يوثق القصص الفائزة في مسابقة الإمام العسكري (عليه السلام)
(نوافذ).. إصدار أدبي يوثق القصص الفائزة في مسابقة الإمام العسكري (عليه السلام)


















