


 الفيزياء الكلاسيكية
الفيزياء الكلاسيكية
 الكهربائية والمغناطيسية
الكهربائية والمغناطيسية
 علم البصريات
علم البصريات
 الفيزياء الحديثة
الفيزياء الحديثة
 النظرية النسبية
النظرية النسبية
 الفيزياء النووية
الفيزياء النووية
 فيزياء الحالة الصلبة
فيزياء الحالة الصلبة
 الليزر
الليزر
 علم الفلك
علم الفلك
 المجموعة الشمسية
المجموعة الشمسية
 الطاقة البديلة
الطاقة البديلة
 الفيزياء والعلوم الأخرى
الفيزياء والعلوم الأخرى
 مواضيع عامة في الفيزياء
مواضيع عامة في الفيزياء|
Read More
Date: 22-3-2016
Date: 24-3-2016
Date: 7-12-2016
|
Dye lasers
Many organic dyes can exhibit a pronounced luminescence which covers a large range of wave-lengths in the visible range of the spectrum. A dye molecule which is quite often used for lasers is Rhodamine 6G. Its molecular structure is shown in fig. 1.1. The optical transitions are caused by electrons. We have to distinguish between two kinds of excitations. In one case the spin of the electron in the excited level is opposite to that of the remaining

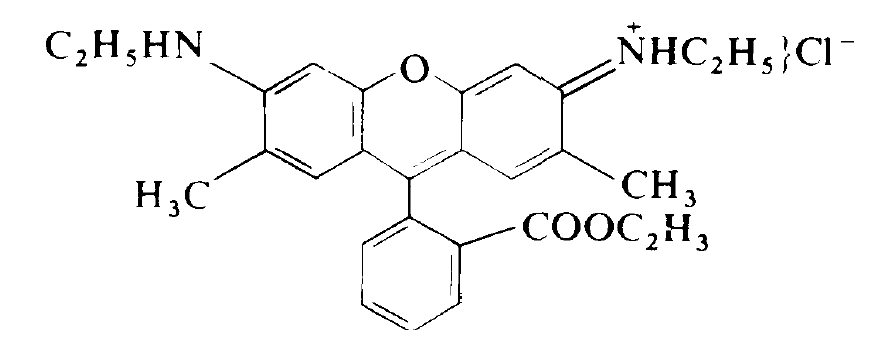
Fig. 1.1. Molecular structure of Rhodamine 6G.
molecule so that a total spin equal 0 results. We call these states S-states (singulet states). In the second case the spin of the excited electron is parallel to that of the remaining molecule. The total spin equals 1 and we are speaking of triplet states. Both kinds of states are further split due to molecular vibrations. The levels are represented in fig. 1.3 by heavy lines. Finally a still finer splitting exists. It stems from the rotation of the molecules which, according to quantum theory, is quantized. In usual experimental setups the dye molecules are in solution. The dye molecules are excited by
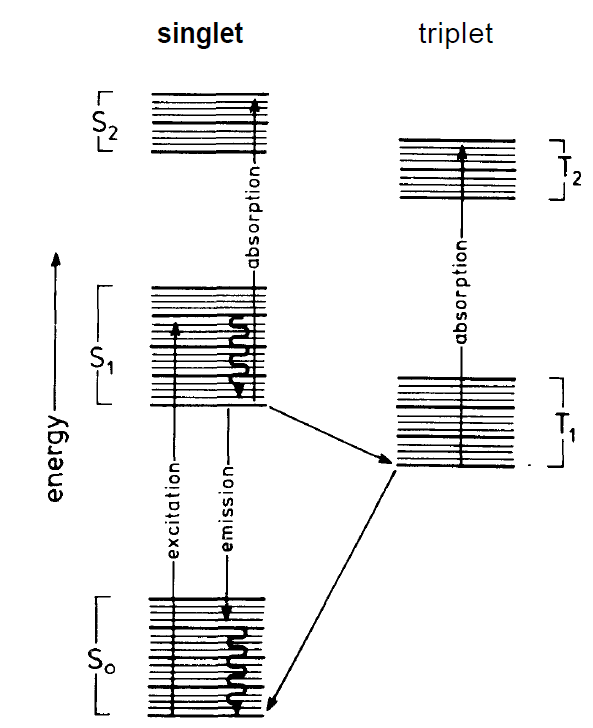
Fig. 1.2. Energy level scheme of a dye. On the left-hand side the groups of the singlet states are plotted which are further split due to oscillations and rotations (compare text). On the right-hand side the triplet states are plotted.
other lasers, e.g. the argon laser, whereby a transition from the group S0 into the group S1 occurs. This excitation is followed by a fast recombination to the lowest level of group S1. From there the optical transition in one of the states of group S0 occurs. Besides this optical recombination a transition from S, to T1 happens with a relatively small transition rate. Because the optical transition from T1 into the ground state of S0 is forbidden, the states T, are long living. Unfortunately the absorption frequency of the transition from T1 to T2 coincides with the emission frequency from S1 to S0. As a consequence the emitted laser light is strongly reabsorbed so that laser action is quickly suppressed. Therefore in such a case only laser light pulses can be emitted. However, by adding new substances to the solution a quick recombination of the states T1 can be achieved so that reabsorption is suppressed. Besides Rhodamine 6G there are a number of further organic dyes showing laser action. By a combination of different kinds of dye molecules a range of wave-lengths from 430 till 800 nm can be covered. On account of their broad luminescence lines, organic dyes are particularly well tunable. Tuning can be achieved, for instance, by reflection gratings.
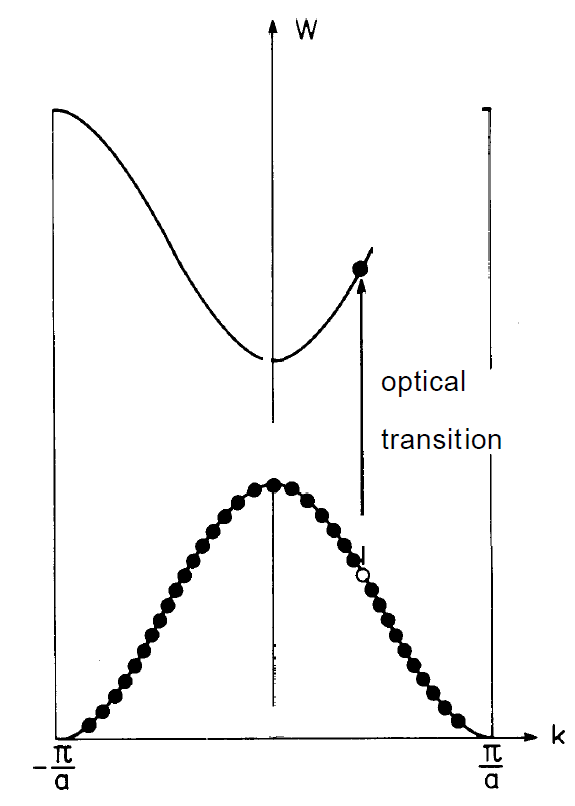
Fig. 1.3. The optical transition of an electron from the valence band into the conduction band takes place under conservation of the k-wave vector, i.e. vertically.



|
|
|
|
"إنقاص الوزن".. مشروب تقليدي قد يتفوق على حقن "أوزيمبيك"
|
|
|
|
|
|
|
الصين تحقق اختراقا بطائرة مسيرة مزودة بالذكاء الاصطناعي
|
|
|
|
|
|
|
قسم شؤون المعارف ووفد من جامعة البصرة يبحثان سبل تعزيز التعاون المشترك
|
|
|